 Air - Molecular Weight and Composition - Dry air is a mixture of gases where the average molecular weight (or molar mass) can be calculated by adding the weight of each component.Acetone - Thermophysical Properties - Chemical, physical and thermal properties of acetone, also called 2-propanone, dimethyl ketone and pyroacetic acid.Material Properties - Material properties of gases, fluids and solids - densities, specific heats, viscosities and more.See also Physical data for hydrocarbons, Physical data for alcohols and carboxylic acids, Physical data for organic nitrogen compounds and Physical data for organic sulfur compounds To calculate the molecular weight of ethanol, the molecular weight of each atom in the molecule is summed: The atomic weights of the elements found in organic substances are C = 12.011, H = 1.008, S = 32.065, O = 15.999, and N = 14.007.Įxample: The molecular weight of ethanol (C 2H 5OH) The molecular weight of a pure compound is determined from its chemical formula and the atomic weights of its elements. For this reason, in many cases the unit for the molecular weight is not mentioned however, one must realize that it is not a dimensionless parameter. Molecular weight is represented by the same number in all unit systems regardless of the system used. In the SI system the unit of M is and in the English system the unit is, while in the cgs system the unit of M is. It does not store any personal data.The molecular weight of a substance, also called the molar mass, M, is the mass of 1 mole of that substance, given in M gram. The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. 
The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". 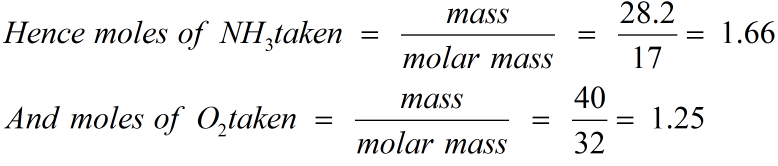
These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a characteristic pungent smell. It is essential for many biological processes and has various industrial applications.Īmmonia is a compound of nitrogen and hydrogen with the formula NH 3. Ammonia is irritating to the skin, eyes, nose, throat, and lungs. The structure of NH3 is not planar, but a tetrahedron, and the nitrogen is on one apex, the center of negtive charge is not the same position as the center of positive charge, so NH3 os polar.Īmmonia is a colorless inorganic compound of nitrogen and hydrogen with the formula NH3, usually in gaseous form with a characteristic pungent odor. Classified as a metalloid, Polonium is a solid at room temperature. Polonium is a chemical element with symbol Po and atomic number 84. 
Therefore, the molar mass = 15.9994 g/molĮxample: Molecular mass of Ca(OH)2 = 74 atomic mass units It can also be expressed as kilogram per mole….Molar mass: Difference between Molar mass and Molecular massĮxample: Mass of 1 mole of oxygen is 15.9994 grams. What is the difference between molar mass and molecular mass?Įxpression of molar mass is grams per mole. The unit used to measure is grams per mole. 
The molar mass, also known as molecular weight, is the sum of the total mass in grams of all the atoms that make up a mole of a particular molecule. It is a polar molecule and is highly associated because of strong intermolecular hydrogen bonding. The ammonia molecule has a trigonal pyramidal shape with the three hydrogen atoms and an unshared pair of electrons attached to the nitrogen atom. What is the mass of NH?Īmmonia is a compound of nitrogen and hydrogen with the formula NH3. This means that its molar mass will be the sum of the molar mass of one nitrogen atom and three times the molar mass of a hydrogen atom. 4 What is the difference between molar mass and molecular mass?ġ7.03052 g/mol. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
